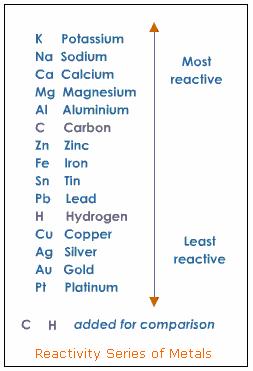
#Reactivity series series#
A metal in the reactivity series below hydrogen will not react with dilute acids. The more reactive the metal, the quicker the reaction. Magnesium, for example, reacts quickly with dilute hydrochloric acids Reaction of metals with dilute acids:Ī salt and hydrogen are generated when a metal combines with a dilute acid. The relative resistance to oxidation of a metal is proportional to its position in the reactivity series in general, the lower a metal's position in the reactivity series, the stronger its resistance to oxidation. It loses oxygen in order to produce hydrogen. Magnesium is oxidised, gaining oxygen to form magnesium oxide, while water is reduced in this reaction. When steam is run over hot magnesium, however, a strong reaction occurs: This shields the metal and prevents it from reacting. When magnesium is first added to water, it reacts slowly, but a coating of insoluble magnesium hydroxide forms. Periodic Trends in Ionisation Enthalpy of Elements.Modern Periodic Table Modern Periodic Law.Rutherfords Model of Atoms and Its Limitations.Electronic Configuration of First 30 Elements.The surface of the iot naturally generates a thin coating of aluminium oxide, which keeps water away from the metal beneath. Aluminum is unique in that it is a reactive metal that does not corrode when exposed to water.

The more reactive the metal, the faster the reaction. Sodium, for example, reacts quickly with cold water:

Reaction of metals with water:Ī metal hydroxide and hydrogen are generated when it reacts with water. It also contains information on metal reactivity in the presence of water and acid. The reactive series tells you whether or not a metal may displace another in a single displacement reaction. We already know that a metal activity series is a list of metals arranged in declining order of their reactivities. Important uses of Activity or reactive series: This statement helps to learn the reactive series of metals. Please stop calling me a careless zebra instead try learning how copper saves gold. Potassium (Please) Sodium (Stop) Calcium (Calling) Magnesium (Me) Aluminum (A) Carbon (Careless) Zinc (Zebra) Iron (Instead) Tin (Try) Lead (Learning) Hydrogen (How) Copper (Copper) Silver (Saves) Gold (Gold) The short trick to remembering reactivity series: NCERT Exemplar Class 11 Chemistry Solutions Chapter 3 Classification of Elements and Periodicity in Properties.NCERT solutions for Class 11 Chemistry Chapter 3 Classification of Elements and Periodicity in Properties.NCERT notes Class 11 Chemistry Chapter 3 Classification of Elements and Periodicity in Properties.Other factors may, however, come into play as metal potassium can be prepared by reducing sodium potassium chloride to 850 ° C.Īlthough sodium in the reactivity series is less than potassium, the reaction can continue since potassium is volatile and the mixture is distilled.In a reactive series of metals, the most reactive element is placed at the top thus, the most reactive metal is Caesium and the least reactive metal is placed at the bottom of the reactive series of metals thus, the least reactive metal is platinum. Likewise, the removal of titanium from tetrachloride can be achieved using magnesium, which in the end forms magnesium chloride: For instance, iron(III) oxide is decreased into iron and aluminium oxide is converted in this process. (Ti is approximately the same level as Al in the reactivity series).

This is used for the thermite reaction for the production of small amounts of metallic iron and for the preparation of titanium by kroll process Generally, any of the metals that are lesser in the reactivity series can be replaced by metal: higher metals reduce lower metal ions. An iron nail in a copper sulphate solution can soon change colour as metallic copper is coated with the iron(II) sulphate.

